Homeostatic Mechanisms, Acids&Bases, Water
What is Homeostasis?
It's the tendency for organisms to maintain a stable internal environment
Fluctuating environments, inside and outside our body, make it difficult for organisms to maintain their stable/optimum environments. Hence, our bodies are constantly trying to maintain these stable internal conditions by physiologically starting and stopping feedback cycles.
Examples:
-
7.4 blood pH, 120/80 blood pressure, 37 degrees Celsius body temperature, 0.1% blood glucose levels
Negative Feedback Cycle
1. A STIMULUS disrupts homeostasis (stability) in the body
2. RECEPTORS throughout the body detect changing internal conditions (ie. instability in the body) and send a signal to the CONTROL CENTER (usually the brain).
3. The REGULATORY CENTER, usually the brain directs the body to behave in a certain way to regain normalcy/homeostasis in the body.
4. An ADAPTIVE RESPONSE is sent out in order to help our body regain homeostasis.
5. Once NORMALCY is reached the process stops and our receptors detect homeostasis and send a signal to the control center to stop the adaptive response.
HOWEVER, IN A NEGATIVE FEEDBACK SYSTEM LEVELS ARE NEVER EXACTLY NORMAL BUT THEY ARE CONSTANTLY FLUCTUATING.
Examples:
-
hunger, lack of oxygen, irregular body temperature, irregular glucose levels, irregular blood pressure, etc.
Stimulus
Receptors
Normalcy
Regulatory Center
Adaptive Response
CYCLE
Positive Feedback Cycle

This response does not trigger an adaptive response to cancel the stimulus. INSTEAD, IT INCREASES THE STIMULUS UNTIL SOMETHING IS EJECTED FROM THE BODY.
LABOR EXAMPLE:
1. The body stimulates labor to begin when pressure is exerted on the cervix.
2. This pressure triggers sensory nerves in the cervix to send a signal to the pituitary gland.
3. The pituitary gland receives the signal and begins to release Oxytocin, a hormone, into the blood stream.
4. This hormone travels to the uterus through the bloodstream where it causes the uterus to contract harder causing more pressure to be felt on the cervix.
5. This process continues and each time more pressure is put on the cervix which in turn causes more Oxytocin to be released and stronger contractions until the baby is delivered.
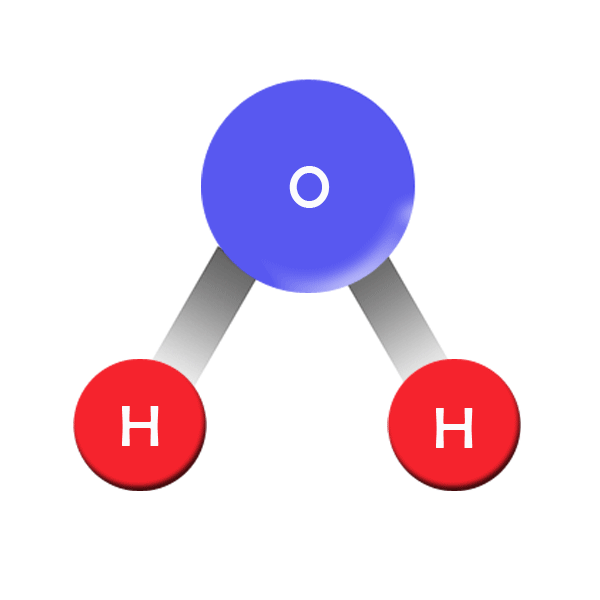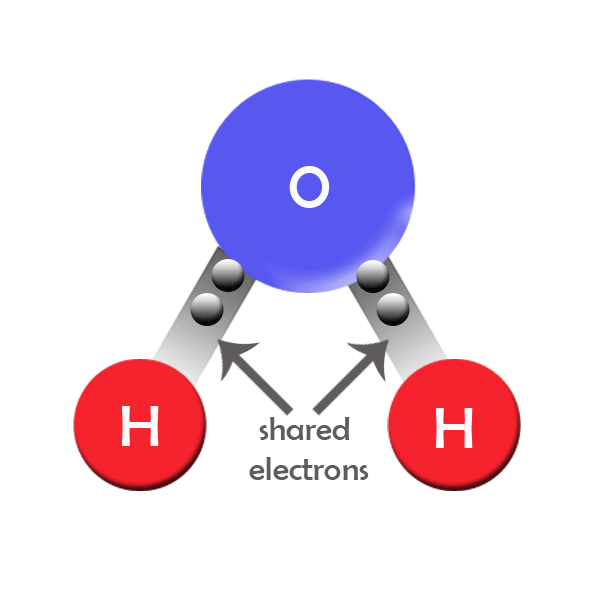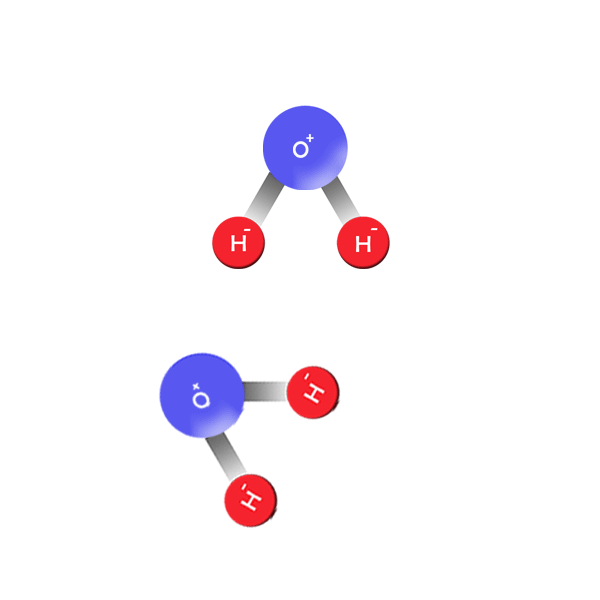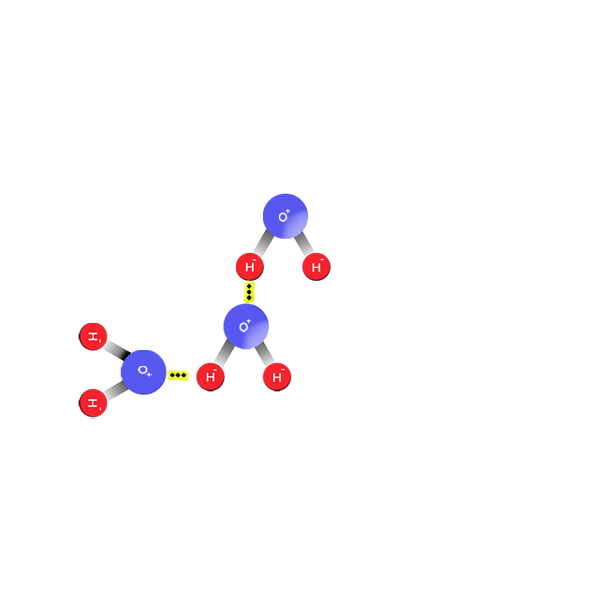
All About Water
ACIDS & BASES

ACIDS
-
Release positive hydrogen ions that can conduct electricity in solutions.
-
They act like acids when dissolved in water and hence, need the subscript 'aq' after them.
-
'Aq' is written to show that acids are dissolved in water
-
acids begin with hydrogen atoms EXCEPT ACETIC ACID (CH3COOH)
Examples: Blueberries, raspberries, blackberries, apples, tomatoes, HCl, etc.
BASES
-
End with OH (hydroxide) in their formulas
-
They act like bases when dissolved in water and hence, also need the subscript 'aq' to show that they are dissolved in water
-
OH- ions can conduct electricity when they dissociate in water
Examples: Bleach, drain cleaner, soap, blood, ammonia, etc.
pH Scale

The pH Scale is a numerical method used to indicate how acidic or basic a compound is.
The pH scale determines how acidic or basic compounds are depending on how many hydrogen ions the solution contains. The more hydrogen ions in a solution the more acidic the solution is and the less hydrogen ions there are the more basic a solution is. A pH meter can also identify acids and bases by determining how many hydrogen ions there are in the solution. Also, indicators, which are compounds that help reveal the pH of solutions by usually changing color, help determine if a solution is acidic or basic.
Examples of Indicators: Litmus Paper, Phenolphthalein, Methyl Red, etc.
Neutralization & Buffers
NEUTRALIZATION:
-
When a solution with a high hydrogen ion concentration mixes with a solution that has a high hydroxide ion concentration then these concentrations cancel each other out
-
When there is an even number of hydrogen and hydroxide ions in a solution that solution is considered to be neutral with no charge.
Neutralization Reaction Example:
H+(aq) + OH-(aq) ---> H2O(l)
BUFFERS:
-
Are chemicals that can take in excess hydrogen or hydroxide ions to decrease or increase the pH
-
they allow our body to maintain homeostasis by keeping our pH constant
-
Without a constant pH of 7.4 in our bodies enzymes would begin to break down and this would be very dangerous
-
Buffers work by dissociating ions so the pH will either increase or decrease
pH SCALE:
THE pH SCALE:
-
MORE H+ ions = Acidic solution with low pH
-
MORE OH- ions = Basic solution with high pH
-
Acids have a pH less than 7
-
Bases have a pH greater than 7
-
Neutral solutions have a pH of 7
-
The pH scale is logarithmic meaning each number on the scale represents a difference of 10